Asymmetric cell division is the process by which a mother cell divides to generate two daughter cells that adopt distinct cell fates. This process is essential to generate cellular diversity in multicellular organisms. Both cell signaling and the asymmetric segregation of intracellular proteins are likely required to specify distinct daughter cell fates during asymmetric cell division. Do all organisms use similar signaling pathways? Are asymmetrically localized determinants conserved between organisms or cell types? How does an asymmetrically localized factor ultimately lead to differential gene expression necessary for cell fate determination?
To address these questions, the Hawkins lab utilizes the nematode Caenorhabditis elegans. C. elegans is ideally suited to investigate the mechanisms controlling asymmetric cell division. In the hermaphrodite, all 302 neurons arise from asymmetric cell divisions. Since the entire cell lineage is known, the timing, location and polarity of cell divisions can be analyzed at the resolution of single cells. In addition, a wealth of molecular and genetic tools also makes C. elegans a powerful system in which to identify and analyze genes involved in asymmetric cell division.
Specifically, we are focusing on asymmetric divisions that occur during embryogenesis in the lineages that generate the two sensory neurons in the tail, PHA and PHB. Both the PHA and PHB neurons are born during embryogenesis and have similar axonal and dendritic projections. Despite these similarities, these two neurons are derived from distinct lineages.
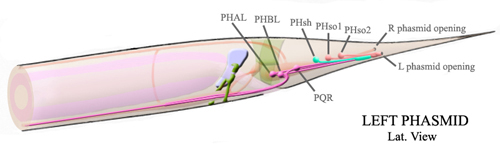
http://www.wormatlas.org/handbook/hypodermis/Phasmidimagegallery.htm
Using specific GFP reporters we can analyze the productions of these two neurons.

Production of the PHA neuron
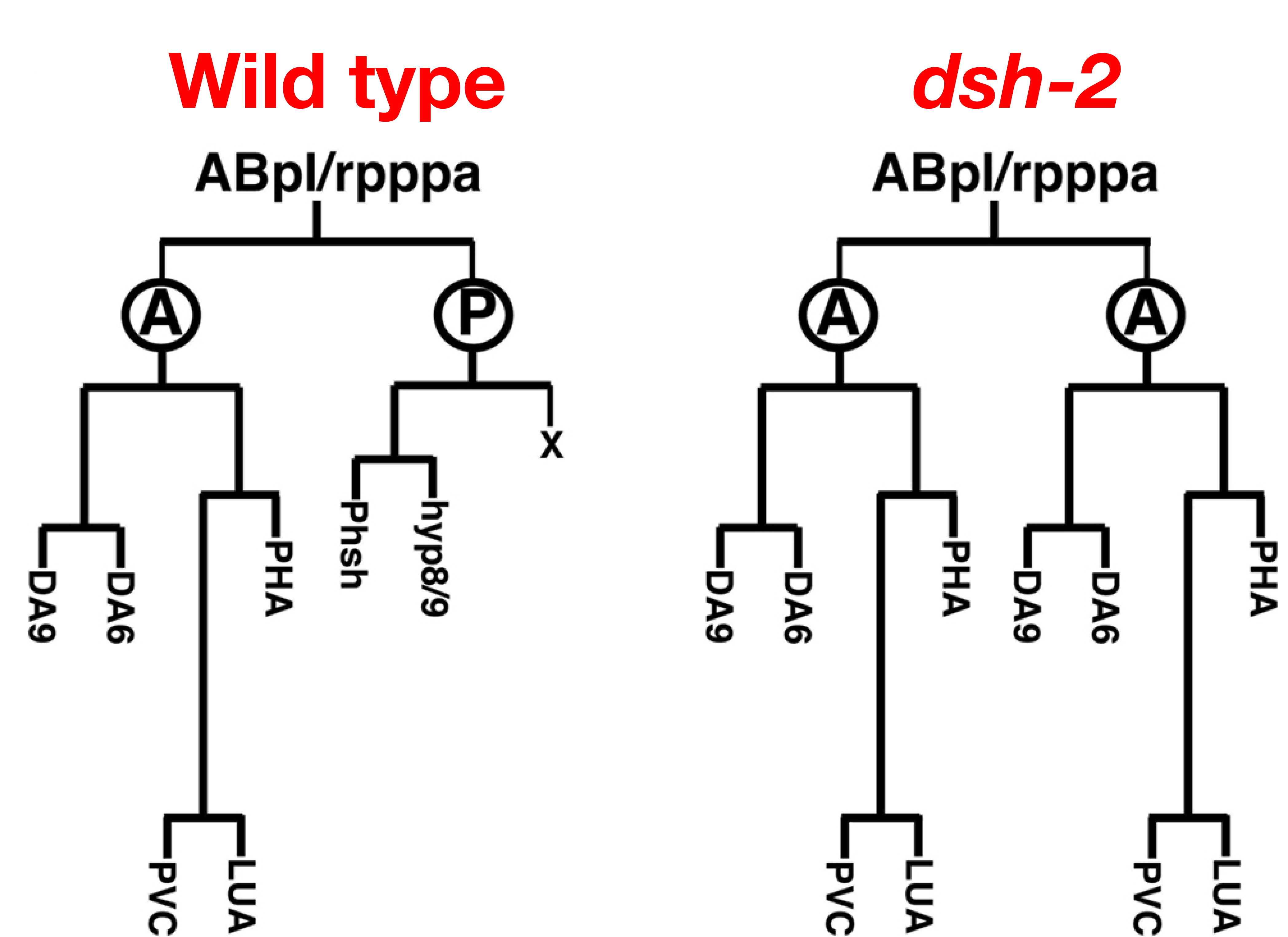
In wild type embryos, a neuroblast divides asymmetrically to generate distinct anterior and posterior daughter cells. The anterior daughter cell divides further to generate 5 neurons, one of which is PHA. The posterior daughter cell produces only non-neuronal cells. We have shown that mutations in both a C. elegans dishevelled homolog (dsh-2) and a frizzled (fz) homolog (mom-5) disrupt asymmetric division of the neuroblast. In the mutants, the neurons normally descended from the anterior daughter cell are often duplicated, while the non-neuronal cells produced by the posterior daughter cell are frequently missing.
In wild type embryos, the DSH-2 protein is normally found at the cell cortex, while in mom-5 mutant embryos the protein is often found in the cytoplasm.This suggests that MOM-5 regulates asymmetric cell division by controlling the localization of DSH-2.
Fz and Dsh are key components of several intracellular signaling pathways with the canonical Wnt/b-catenin pathway being the most well studied. Misregulation of the Wnt pathway is one of the most common signaling abnormalities in human cancers. Fz and Dsh. However, Fz and Dsh are also components of a planar cell polarity (PCP) pathway and a Wnt/Ca2+ pathway. We want to identify additional pathway components that participate with DSH-2 amd MOM-5 in regulating asymmetric neuroblast division in C. elegans to ultimately understand the molecular mechanism underlying this process. To accomplish these goals we are taking several approaches.
1. Is there a Wnt involved in polarizing dividing neuroblasts? There are five Wnt genes in C. elegans.Using a combination of mutants and RNAi, we have been testing the requirements of all five Wnts.Thus far, we have uncovered a requirement for cwn-1.
2. Are additional asymmetric cell divisions affected in dsh-2 mutants? Using a combination of cell specific GFP reporters we have identified at least 14 additional cells whose production is affected in dsh-2 mutants.
3. Which pathway does DSH-2 function in to regulate asymmetric cell division? Initially to address this question, we performed RNAi experiments to test the requirements of all the C. elegans Wnt and PCP genes. Unfortunately, we could not uncover a role for the ~ 40 genes tested. Therefore, we have performed a genetic screen to isolate dsh-2 suppressors. This screen was highly successful. We isolated over 80 suppressors. The majority of the suppressors are dominant and therefore could be activating mutations in downstream pathway components. In addition, we have also isolated a handful of recessive suppressors. We are currently genetically mapping of subset of these suppressors with the ultimate goal of identifying the corresponding genes.
Production of the PHB neuron
In the lineage that generates PHB, the HSN/PHB neuroblast divides asymmetrically to generate a smaller anterior daughter cell that dies and a larger posterior daughter cell, the HSN/PHB precursor. The HSN/PHB precursor then divides to produce an PHB and HSN neuron. In ham-1 mutants, the anterior daughter cell can inappropriately survive and is transformed into a second HSN/PHB precursor, resulting in PHB and HSN neuron duplications . A close examination of the HSN/PHB neuroblast division reveals the daughter cell sizes are reversed; the anterior daughter cell is often larger than its posterior sister. Thus, we propose that ham-1 controls asymmetric spindle positioning to influence the position of the cleavage plane. This could affect the inheritance of cell fate determinants leading to cell fate transformations.
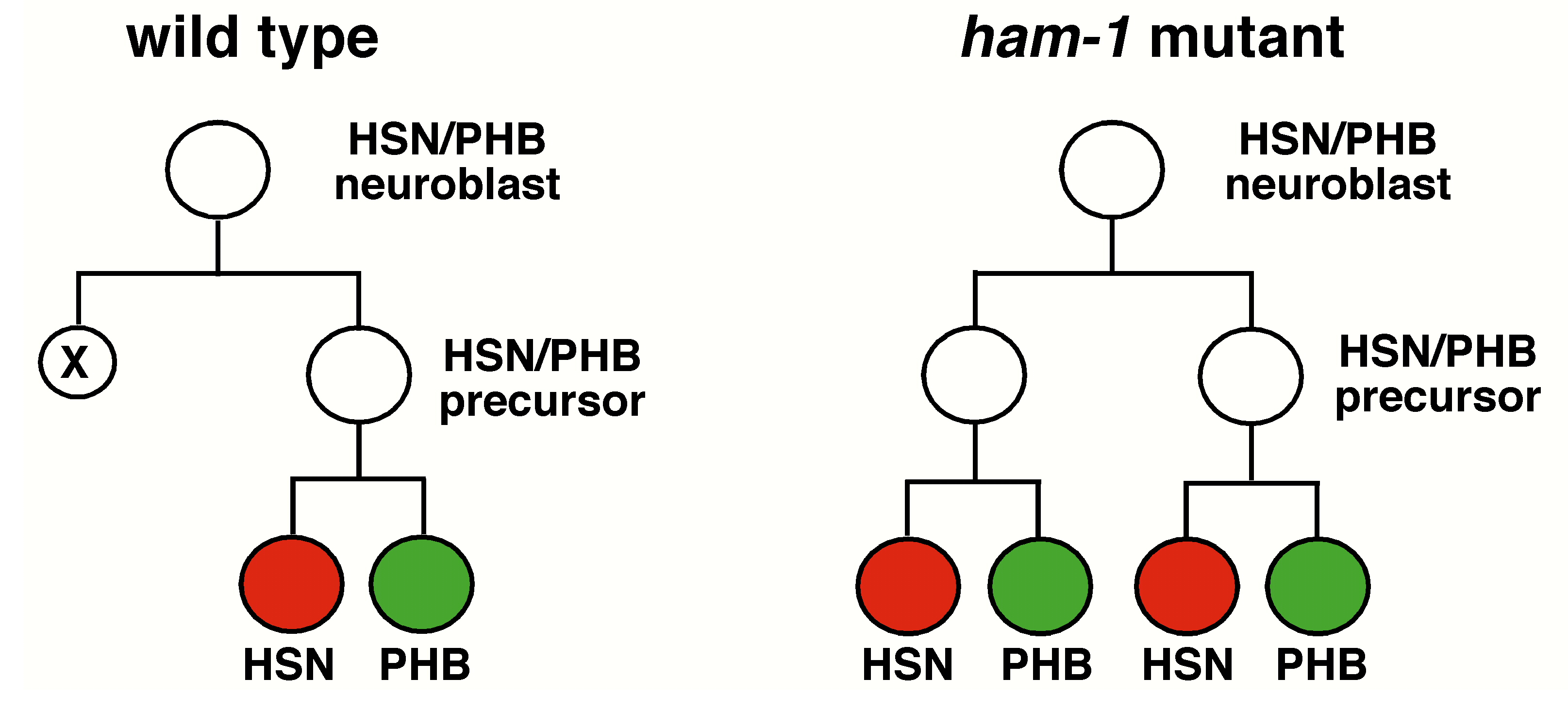
ham-1 encodes a membrane associated protein with no recognizable domains that is asymmetrically localized in many mitotically active cells during embryogenesis; including the HSN/PHB neuroblast
.jpg)
We are taking two main approaches to understand the mechanism by which HAM-1 polarizes asymmetrically dividing cells.
1. Since HAM-1 is a novel protein we are performing a deletion analysis to identify regions of the protein necessary for its asymmetric localization and function. These experiments will identify a novel asymmetry domain.
2. We are also investigating a potential role for Wnt signaling in asymmetric divisions requiring HAM-1.
By two hybrid analysis, we have shown that HAM-1 physically interacts with DSH-1 and MIG-5, two other C. elegans dishevelled homologs.Although RNAi of DSH-1 or MIG-5 does not produce any Ham-1 like phenotypes we have identified a role for two other conserved Wnt pathway components. Loss of either pop-1 (the C. elegans LEF/TCF transcription factor) or apr-1 (the C. elegans APC homolog) function results in phenotypes opposite to HAM-1. Thus, we are further investigating if HAM-1 may function in a Wnt signaling pathway to regulate asymmetric neuroblast division.
|











.jpg)